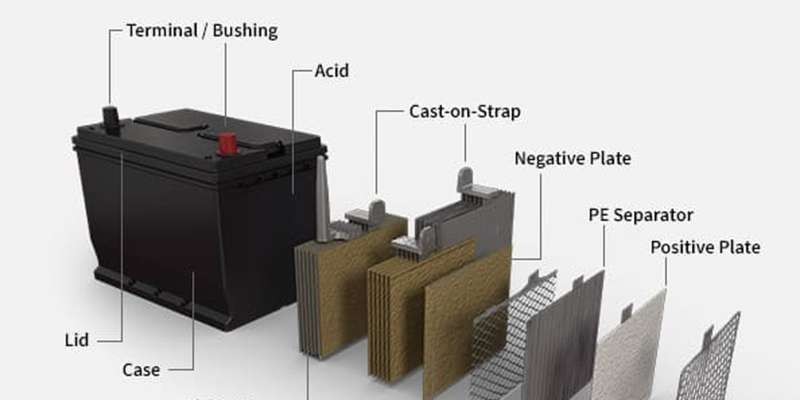
The car battery is the unsung hero of your vehicle. Whether you drive a traditional fuel engine car or a modern electric vehicle, a functioning battery is crucial to get you started. It’s more than just a box of energy; it’s a sophisticated component that stores and releases power to set your car in motion and keep its electrical systems running. In this comprehensive guide, we will delve deep into the essential Car Battery Parts, explaining their roles and how they contribute to your vehicle’s overall performance.
What Exactly is a Car Battery?
At its core, a car battery is a device that converts chemical energy into electrical energy. Specifically, a car battery is a rechargeable, wet-cell battery designed to provide a surge of power to start the engine and then continuously supply electricity to the vehicle’s electrical components. Regardless of the type of car, the fundamental function remains the same: to power the ignition and support the vehicle’s electrical demands.
When you turn the key or press the start button, the battery immediately delivers the electric current necessary to initiate the internal combustion engine or power the electric motor. Beyond starting the engine, the battery is also responsible for powering a wide array of electrical systems, including headlights, interior lights, radio, windshield wipers, and more.
Components of a Typical Car Battery: A Detailed Breakdown
A typical car battery is housed within a robust battery case, protecting its internal components from the harsh under-hood environment. Let’s explore the key car battery parts that work in harmony inside:
Battery Acid (Electrolyte)
The battery acid, more accurately known as the electrolyte, is a crucial component. In lead-acid batteries, which are common in most gasoline-powered cars, this electrolyte is a solution of sulfuric acid and water, typically ranging from 30% to 50% sulfuric acid concentration. This precise mixture is critical because it facilitates the chemical reactions that generate the electrical current. The purity of the acid is paramount; contaminants can negatively impact its effectiveness in producing power.
The interaction between the acid and the battery plates (discussed below) is what generates the voltage needed to start your car. A weak battery, often due to electrolyte degradation or insufficient charge, will struggle to produce the necessary voltage.
Battery Terminals
Battery terminals are the vital connection points that bridge the battery to your car’s electrical system. Think of them as gateways for electrical energy to flow in and out. Every car battery has two terminals:
- Positive Terminal (+): Usually marked with a plus sign (+) and often larger than the negative terminal. It’s the point where electrical current flows out of the battery to power the car.
- Negative Terminal (-): Marked with a minus sign (-) and often smaller. It’s the point where electrical current flows back into the battery, completing the circuit.
Clean and secure terminals are essential for reliable electrical flow. Corrosion or loose connections at the terminals can impede current, leading to starting problems or electrical malfunctions.
Battery Case
The battery case is the protective outer shell that encases all the internal car battery parts. It’s typically made from durable materials like polypropylene resins in traditional car batteries or aluminum alloys in EV batteries. The case serves several critical functions:
- Protection: It shields the delicate internal components from physical damage, vibrations, heat, and the elements.
- Containment: It safely contains the battery acid, preventing leaks and potential hazards.
- Structural Integrity: It provides structural support, holding all the components in place.
A robust battery case is crucial for extending the lifespan of the battery by minimizing external impacts and maintaining the integrity of the internal components.
Battery Plates (Electrodes)
Battery plates are the workhorses of the car battery, where the electrochemical reactions take place. A typical car battery contains multiple sets of positive and negative plates immersed in the electrolyte.
- Positive Plates (Anode): These are typically made of a lead grid coated with lead dioxide. During discharge (when the battery is providing power), lead dioxide reacts with the sulfuric acid.
- Negative Plates (Cathode): These are made of a spongy lead grid. During discharge, the spongy lead also reacts with the sulfuric acid.
These plates are arranged alternately and are designed to maximize the surface area for chemical reactions, thus increasing the battery’s power output capacity. Cast-on straps connect the plates within each cell, linking them to the battery terminals.
Battery Separators
Battery separators are thin, porous insulators placed between the positive and negative plates. They are typically made of polyethylene or other specialized plastic polymers. Their crucial role is to:
- Prevent Short Circuits: Separators physically isolate the positive and negative plates, preventing them from touching and causing a short circuit.
- Facilitate Ion Flow: While preventing physical contact, separators are designed to be porous, allowing the flow of ions within the electrolyte solution. This ion flow is essential for the electrochemical reactions that generate electricity.
Without effective separators, the battery would quickly fail due to internal shorts.
Working Principles: How Car Batteries Power Your Ride
The magic of a car battery lies in its ability to convert chemical energy into electrical energy on demand. Here’s a simplified look at the working principle:
- Chemical Reaction: When you start your car, a chemical reaction begins between the lead plates and the sulfuric acid electrolyte. This reaction releases electrons, creating an electrical current.
- Electron Flow: These electrons flow from the negative terminal, through the car’s electrical circuits, and back into the positive terminal, powering the starter motor and other electrical components.
- Voltage Regulation: The battery also plays a role in stabilizing the electrical system. It helps regulate voltage, preventing surges and maintaining a consistent electrical supply to protect sensitive car electronics.
It’s important to note that starting a car only utilizes a small percentage of the battery’s capacity. Car batteries are designed to deliver a high current burst for a short duration, primarily for starting, lighting, and ignition (SLI).
Exploring Different Types of Car Batteries
The type of battery in your car can influence its performance and longevity. Here are some common types of auto batteries:
Lead-Acid Batteries
Lead-acid batteries are the most traditional and widely used type, especially in gasoline-powered vehicles. They are known for:
- Cost-Effectiveness: They are generally less expensive than other types.
- High Power Output: They can deliver a significant surge of power for starting engines.
- Reliability: Proven technology with a long history of reliable performance.
Lead-acid batteries come in flooded and Absorbed Glass Mat (AGM) variations, with AGM batteries offering improved performance and spill-proof design.
Lithium-Ion (Li-ion) Batteries
Lithium-ion batteries are becoming increasingly prevalent, especially in electric vehicles and hybrid cars. They offer several advantages:
- High Energy Density: They store more energy for their size and weight compared to lead-acid batteries.
- Longer Lifespan: Typically last longer than lead-acid batteries.
- Faster Charging: Can be charged more quickly.
While initially more expensive, the long-term benefits and performance advantages of lithium-ion batteries are driving their adoption in the automotive industry.
Solid-State Batteries
Solid-state batteries represent the next generation in battery technology. They are characterized by:
- Solid Electrolyte: Replacing the liquid electrolyte with a solid material, enhancing safety and stability.
- Improved Energy Density: Potential for even higher energy density than current lithium-ion batteries.
- Faster Charging and Longer Lifespan: Promising faster charging times and extended lifespans.
Solid-state battery technology is still under development but holds immense potential for revolutionizing electric vehicle performance and range.
Key Functions of Car Batteries: Beyond Just Starting
Car batteries perform several crucial functions beyond simply starting the engine:
Engine Starting Powerhouse
The most fundamental role of the car battery is to provide the initial burst of power to crank the engine and get your car running. Without a functioning battery, starting your car would be impossible.
Powering Electrical Accessories
Even when the engine is off, the battery provides power to certain electrical accessories like interior lights, hazard lights, and the car radio (for a limited time).
Supporting the Alternator
Once the engine is running, the alternator takes over as the primary source of electrical power for the car’s systems. However, the battery works in conjunction with the alternator. The battery provides the initial power, and the alternator recharges the battery and sustains the electrical load while driving.
Voltage Regulation and System Protection
Car batteries also contribute to voltage regulation within the vehicle’s electrical system. They can absorb voltage fluctuations, protecting sensitive electronic components from damage caused by overvoltage.
WayKen: Your Partner for Quality Car Battery Parts
The battery case is a critical car battery part that protects users and ensures the integrity of all internal components. When it comes to manufacturing high-quality battery enclosures, especially for electric vehicles, WayKen is your reliable partner.
At WayKen, we offer expertise in EV battery box design and manufacturing, providing customized solutions to fit your specific vehicle requirements. Beyond battery boxes, we also specialize in manufacturing various car parts, including headlamps, backlights, and other essential components.
Our team of experienced engineers and technicians utilizes cutting-edge manufacturing technologies such as CNC machining, injection molding, and 3D printing to deliver precision-engineered car parts. Simply upload your CAD files, and let us bring your automotive designs to life.
Conclusion: The Importance of Understanding Car Battery Parts
A well-functioning car battery is essential for a reliable vehicle. Understanding the various car battery parts and their functions empowers you to appreciate this vital component and ensure its proper maintenance. From the electrolyte facilitating chemical reactions to the protective battery case, each part plays a crucial role in delivering the power your car needs.
FAQs About Car Battery Components
What are the basic components of a car battery?
The fundamental car battery parts include the electrolyte (battery acid or lithium salt solution), positive plates (anode), negative plates (cathode), separators, terminals, and the battery case. The case is crucial for safety and containment, protecting the internal components.
What is the liquid inside a car battery?
In traditional lead-acid car batteries, the liquid is the electrolyte, often referred to as battery acid, which is a solution of sulfuric acid and water. In lithium-ion batteries, the liquid electrolyte is a solution of lithium salt, such as LiPF6, LiClO4, or LiBF4.